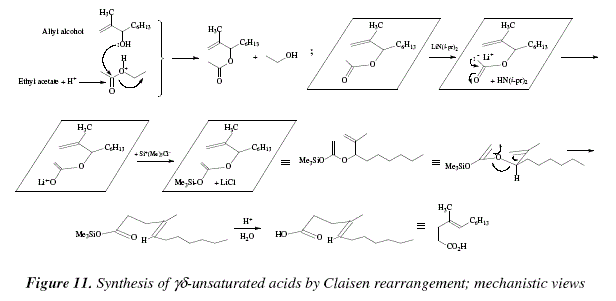
The general structure is r 2 c cr or where r h alkyl or aryl a common subfamily of enol ethers are vinyl ethers with the formula roch ch 2 important enol ethers include the reagent 3 4 dihydropyran and the monomers methyl vinyl ether and ethyl vinyl ether.
Formation of vinyl ethers.
3 2 alkoxide and silyloxide leaving groups.
4 olefination of carbonyl substrates.
Silyl ethers are usually used as protecting groups for alcohols in organic synthesis since r 1 r 2 r 3 can be combinations of differing groups which can be varied in order to provide a.
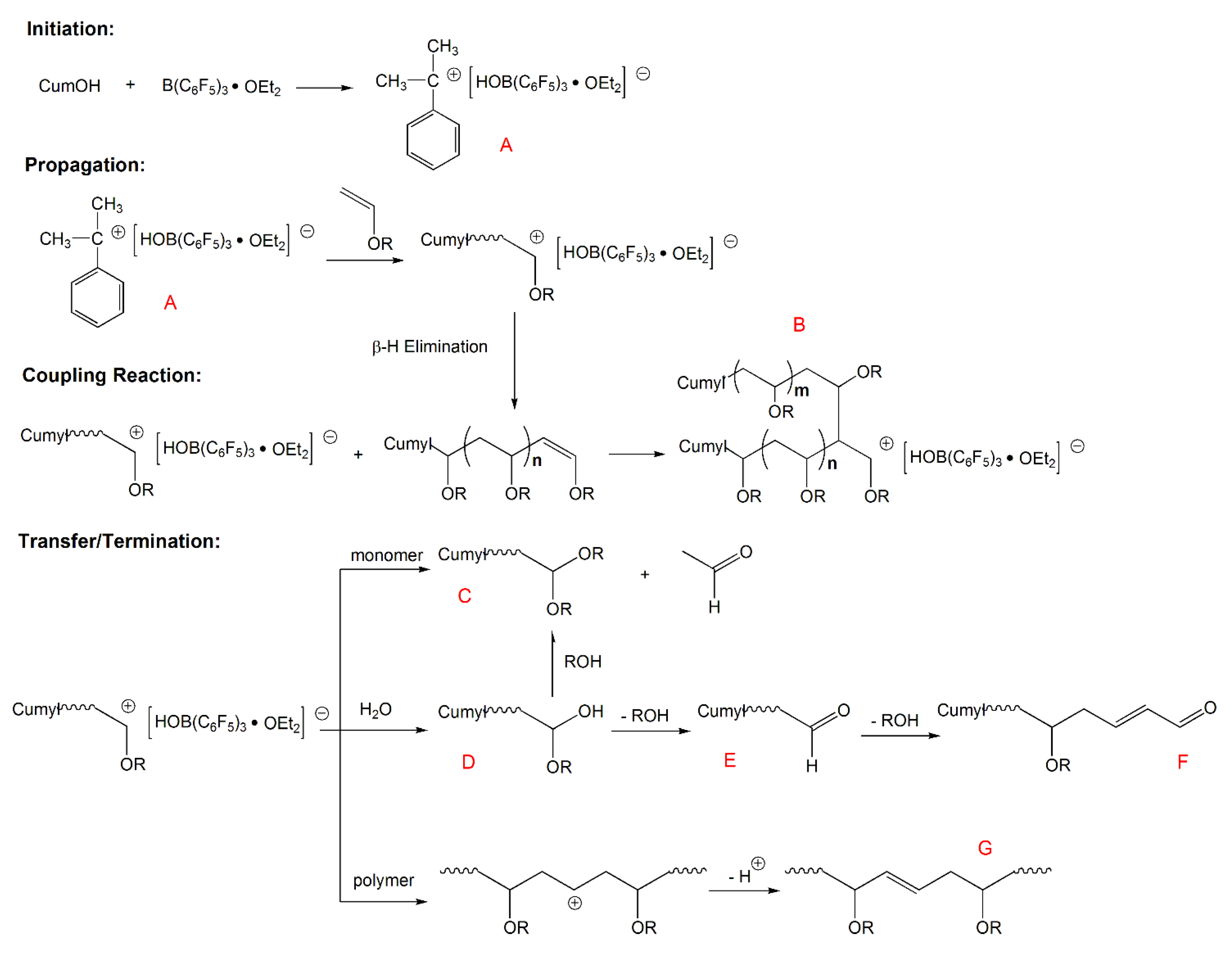
8 21 227 228 the mechanism for chain transfer is shown in scheme 9 for the case of α benzyloxystyrene 15 the driving force for fragmentation is provided by formation of a strong carbonyl double bond.
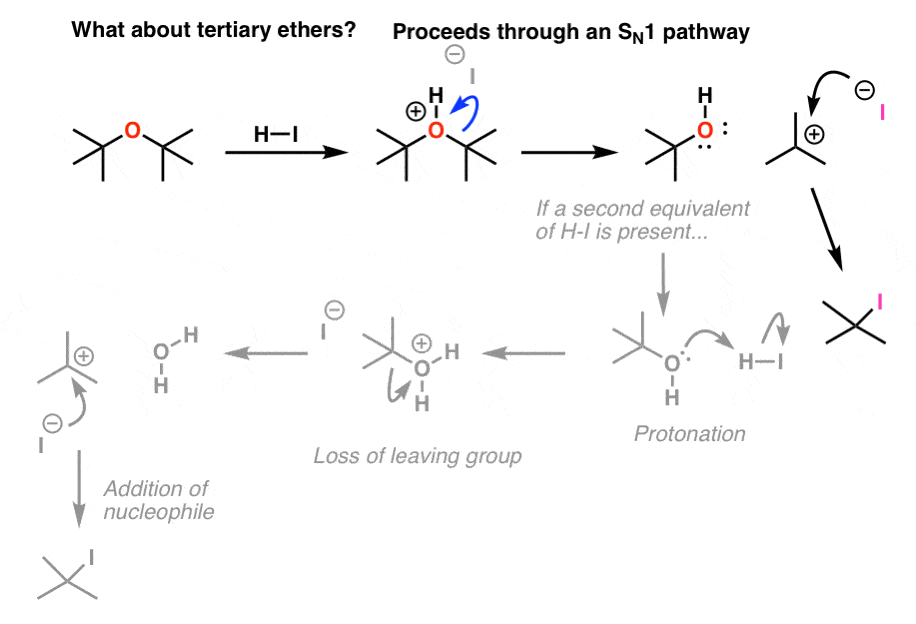
3 1 halide and pseudohalide leaving groups.
It is also important that r is a good radical leaving group.
2 3 acetylenic ether substrates.
These vinyl transfer reactions proceed in good yields with amide carbamate and sulfonamide nucleophiles and the optimal catalyst is dpp pd ococf3 2 dpp 4 7 diphenyl 1 10 phenanthroline.
Sakurai 177 has found that dicobalt octacarbonyl is an excellent catalyst for the reaction equation 67 and that the side reactions occurring through the use of the catalysts mentioned above e g.
Vinyl ethers 1 x ch 2 a o can be very effective addition fragmentation chain transfer agents.
Methyl t butyl ether mtbe is a gasoline additive that boosts the octane number and reduces the amount of nitrogen oxide pollutants.
A stereospecific and stereoselective copper promoted coupling of vinyl pinacol boronate esters and alcohols allows the synthesis of enol ethers in very good yields is compatible with various functional groups and occurs at room temperature.
2 modification of ethers.
Cupric acetate is the copper source and triethylamine buffer is used to prevent protodeboration.
In organic chemistry an enol ether is an alkene with an alkoxy substituent.
Molecular structure is the primary factor relating to a material s potential for hazardous peroxide formation.
It is also used as a volatile starting fluid for diesel engines and gasoline engines in cold weather.
These compounds produce organic peroxides that are significantly less volatile than the solvent in which they are.
Silyl enol ethers have been prepared by reaction of carbonyl compounds with hydrosilanes in the presence of palladium salts rhodium complexes and nickel salts.
Glycol ethers decalin and 2 propanol are shown in group b of the previous section.
Silyl ether formation may be avoided.
Silyl ethers are a group of chemical compounds which contain a silicon atom covalently bonded to an alkoxy group.
Dimethyl ether is used as a spray propellant and refrigerant.
Palladium ii complexes catalyze the formation of enamides via the formal cross coupling reaction between nitrogen nucleophiles and vinyl ethers.
The general structure is r 1 r 2 r 3 si o r 4 where r 4 is an alkyl group or an aryl group.